Ultrapure water is a vital component in several processes, including those frequently used in the electronics, pharmaceutical and medical industries. In order classify water as ultrapure, it must exclusively contain H20 and equal amounts of H+ and OH- ions.
Ultrapure water is the purest type of water that can be produced industrially. The level of contaminants is the minimum possible, whether in terms of minerals, organic elements, or dissolved gases. Ultrapure water is characterized by a neutral pH, a very high resistivity, and no measurable organic content.
Other characteristics of ultrapure water are a TOC <10 ppb and a bacterial count <10 CFU/ml, meaning that ultrapure water contains no bacteria .
As a matter of fact, in the industry, some problems such bacterial contamination and corrosion are solved by ultrapure water.
Ultrapure water is safe for the environment.

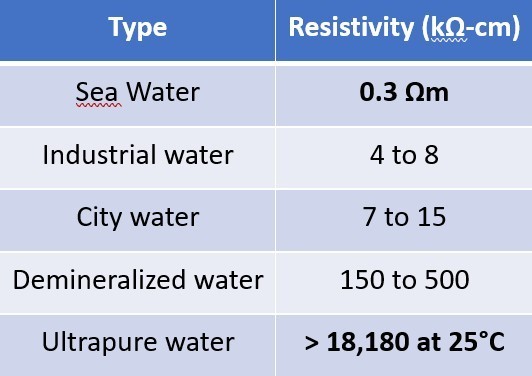
Resistivity is an essential parameter when it comes to electrochemical corrosion.
The very high resistivity of ultrapure water stands out from other types of water and especially seawater.
As opposed to some beliefs, pure water is not a good drinkable water. However, drinking occasionally a glass of ultrapure water will not make you sick.
Living bodies need minerals and other vital elements from drinking water. If you do drink a glass of ultrapure water, you won’t have an immediate bad reaction, but the water will pull some minerals out of your body. As the pure water flows through you, it will attract molecules that should stay there. Essentially, if you only drink pure water, you would lose many important electrolytes. Pure water acts as sponge and soaks up what’s around it, meaning that if you drank nothing but ultrapure water, the water would drink you back.
